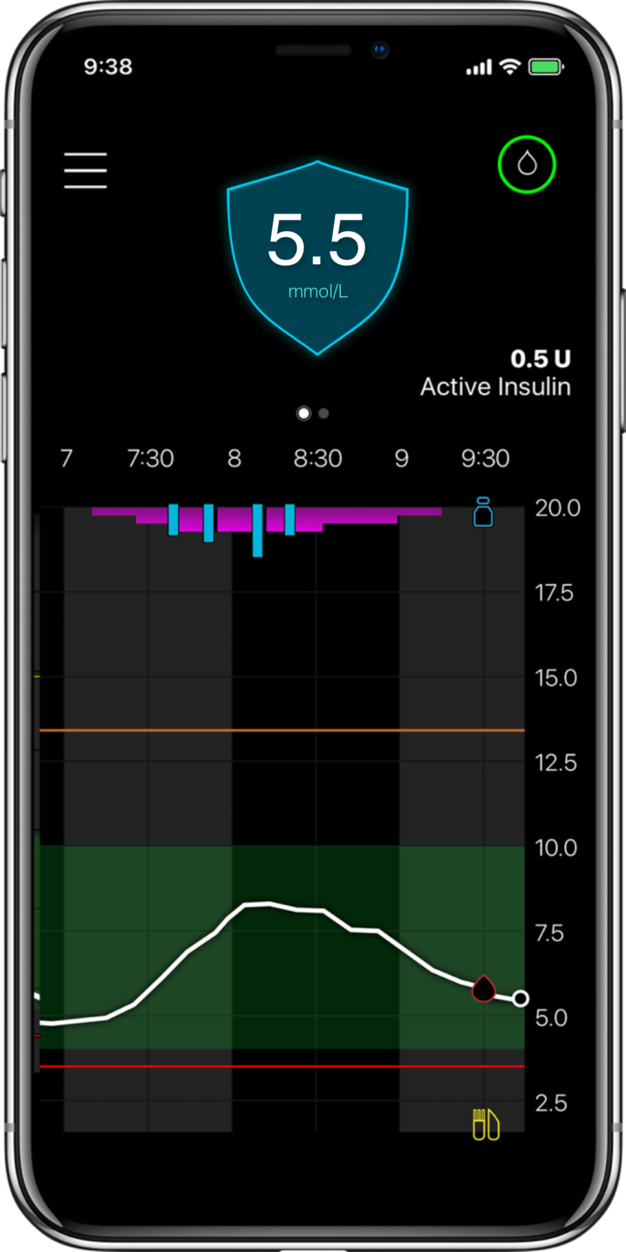
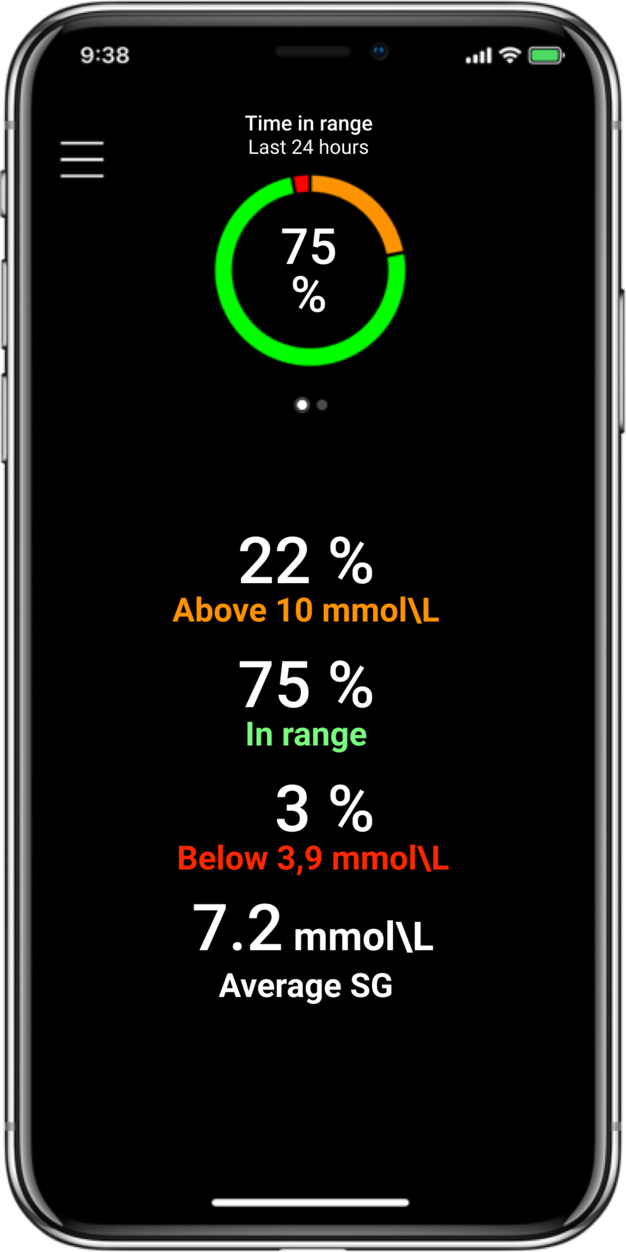
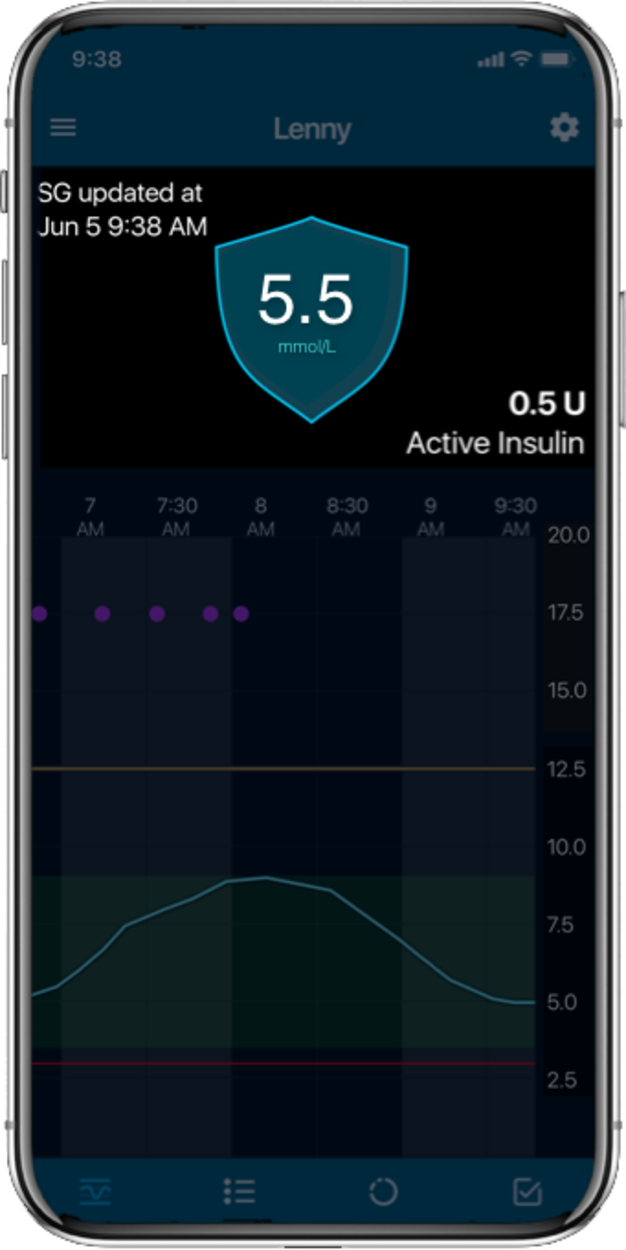
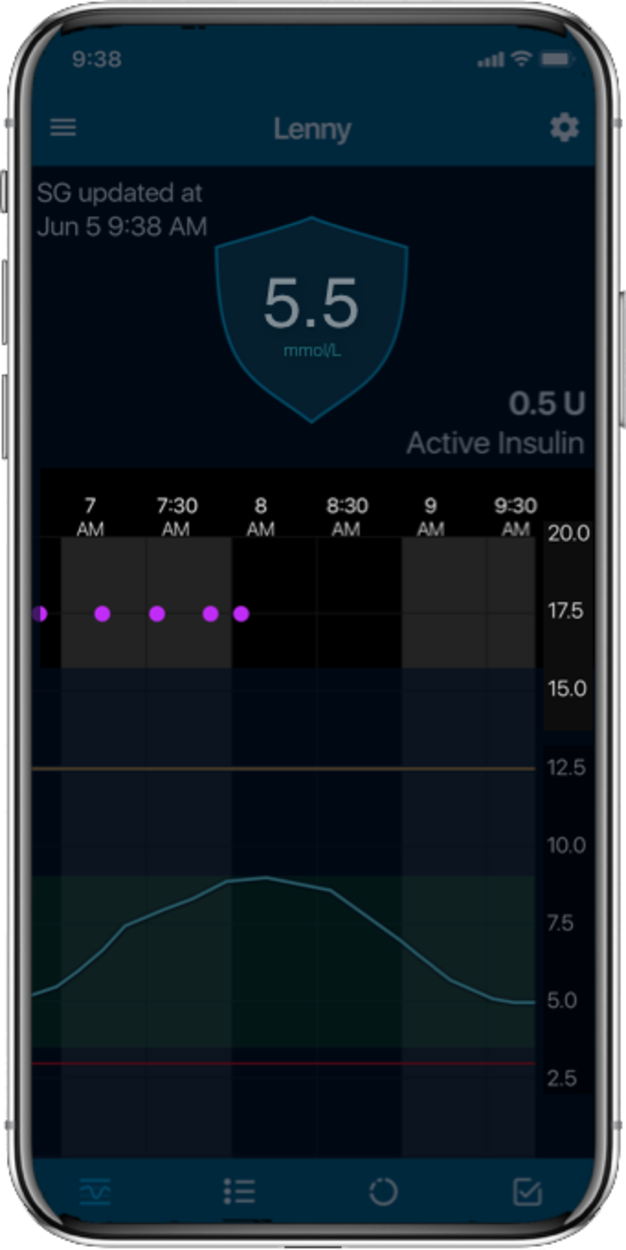
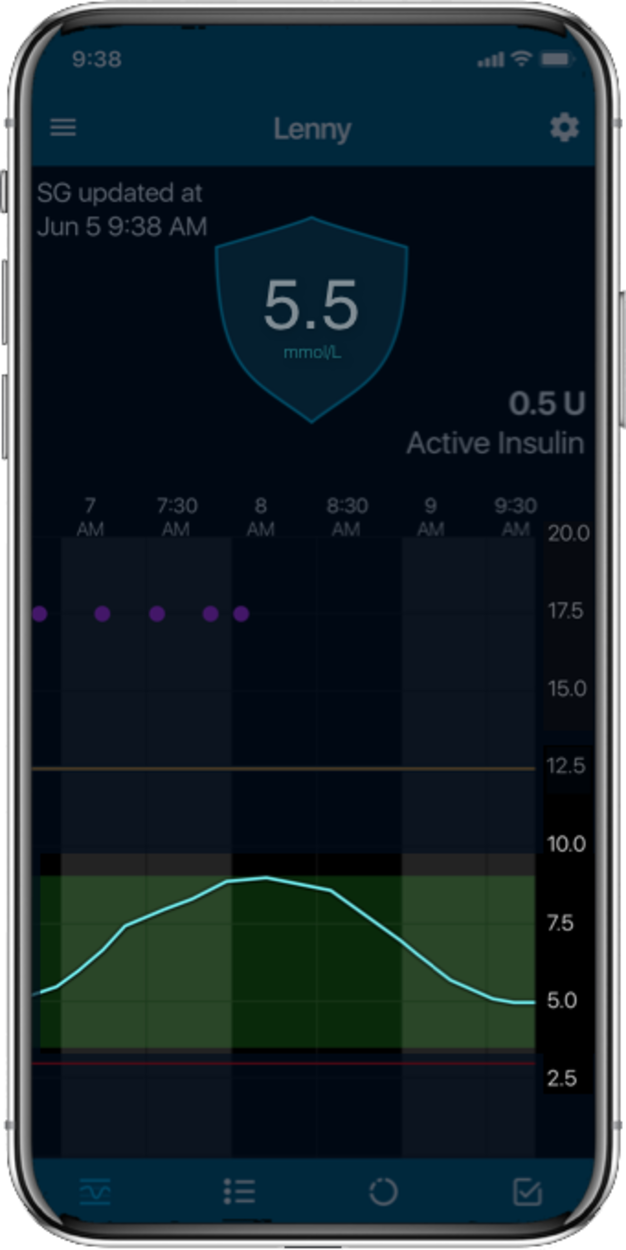
MiniMed™ 780G systemLive life with more
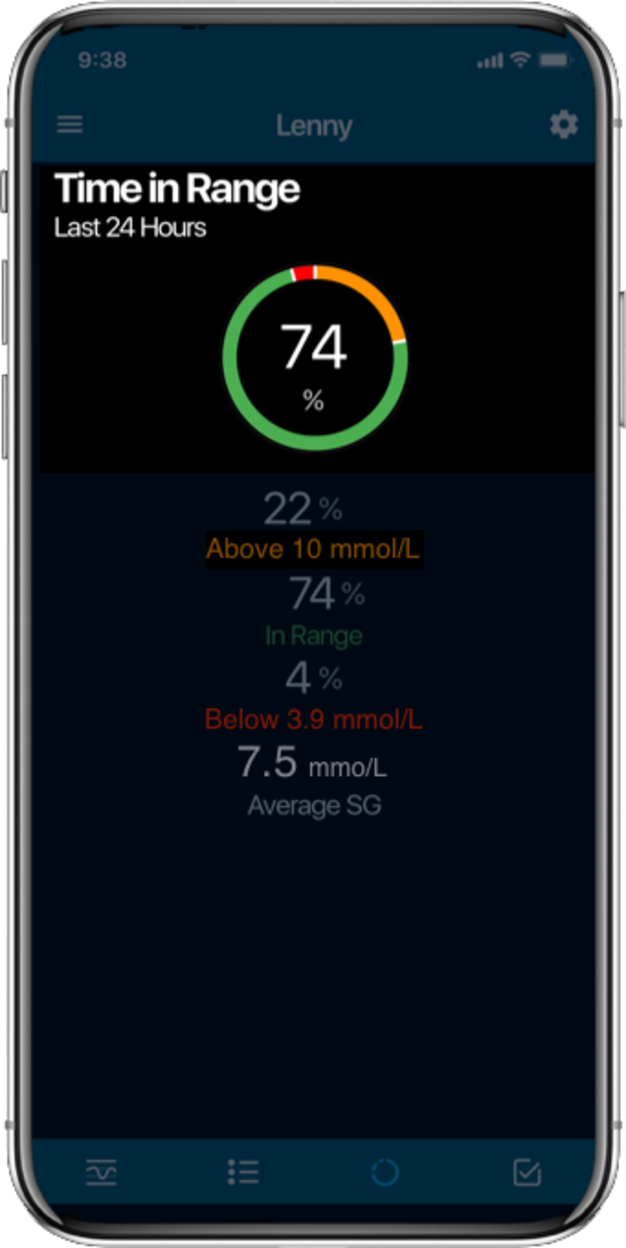
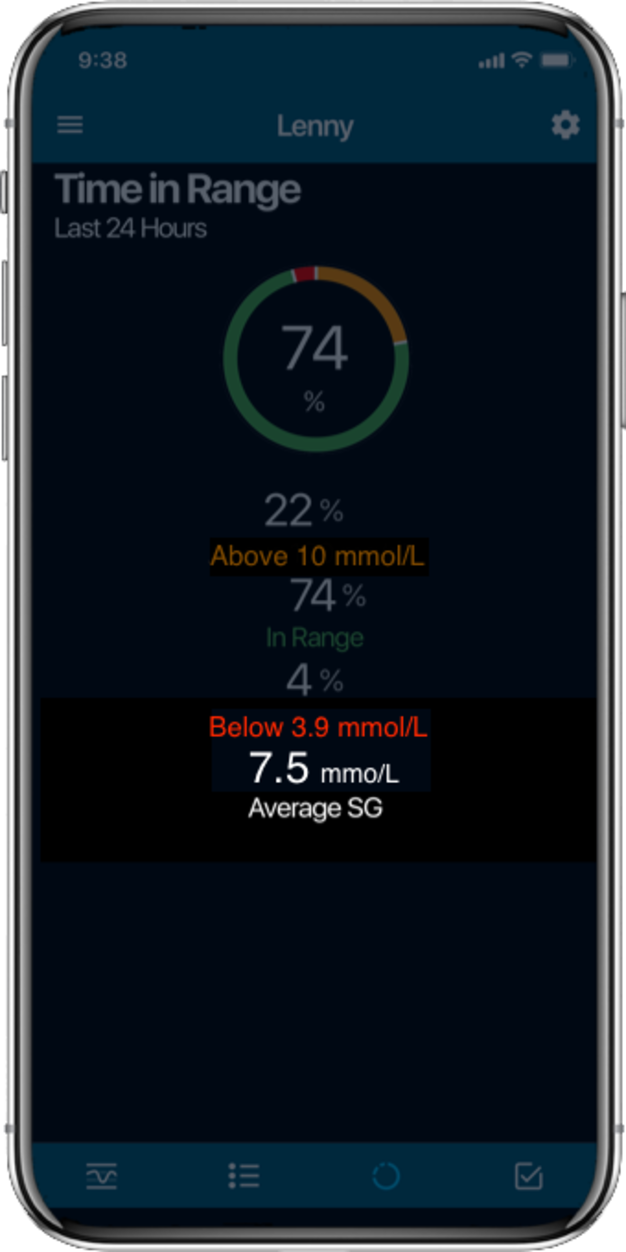
Time in Range1,
For people with type 1 diabetes
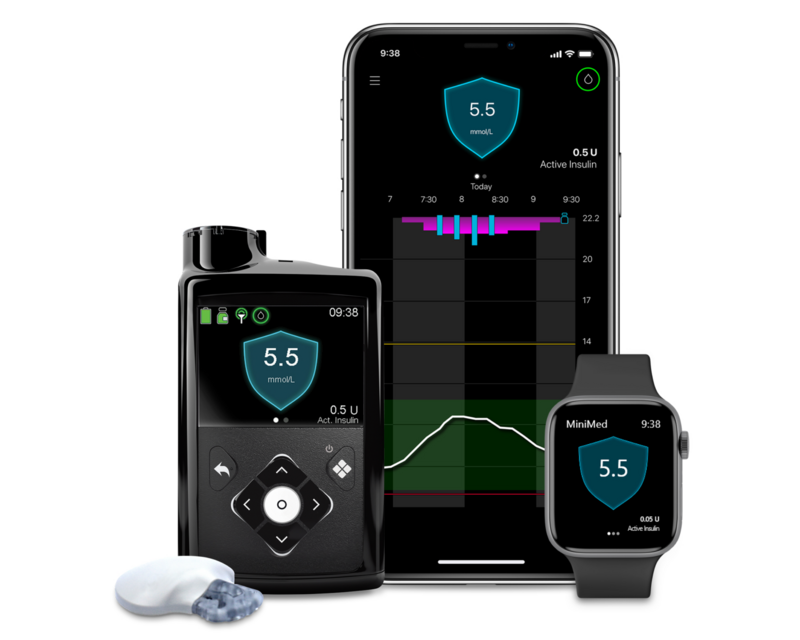
MiniMed™ 780G system
More Time in Range1, , day and night.
The MiniMed™ 780G system
Now available for children with diabetes ages 2 years old+ and women during pregnancy.

Considering an automated insulin pump system?
When choosing an insulin pump, make sure it meets your needs and delivers the best results. Key considerations should include:
- Time in Range (TIR):
The percentage of time your blood glucose levels are within the target range. Higher TIR reduces complications like eye damage (retinopathy), heart and kidneydisease2 . - Burden reduction:
Achieve more while doing less. Your pump should help you to stay in range even when carb counting isn'texact3.
Things you should know about the MiniMed™ 780G
Advanced algorithm
The algorithm adapts to you providing better time in range than other
Wear what you want
Feel free to hide or show the pump depending on your outfit choice.
No daily charging
Unlike other pumps, the 780G doesn't need daily charging.
Freedom to disconnect
Disconnect for up to an hour for activities.
Waterproof design
A tough, easy-to-use pump.
Trusted across Europe
Used by hundreds of thousands, 780G is Europe’s most popular automated insulin pump.
SmartGuard™ technology
Our most advanced insulin pump system
with SmartGuard™ technology
SmartGuard™ technology continuously anticipates your insulin needs, adjusts insulin delivery, and corrects highs
Meal detection
Stronger correction
bolus allowed while
meal is detected
Glucose levels
Automatic adjustments
Auto corrections
Meal detection
For illustrative purposes only.

I can just eat, maybe make a wrong calculation but it will fix it by
itself.
Yonatan
MiniMed™ 780G user

Life is really better with my new
pump.
Talha
6 years old living with type 1 Diabetes using MiniMed™ 780G system.
Now available for children with diabetes 2 years old and above.
Is your mobile phone compatible with Medtronic apps?
| Waterproof capabilities | The pump is waterproof at the time of manufacture and when the reservoir and tubing are properly inserted. It is protected against the effects of being underwater to a depth of up to 2.4 meters (8 feet) for up to 30 minutes. |
| Environmental Conditions | The MiniMed™ 780G insulin pump system is designed to withstand most conditions encountered in your daily life. Pump storage temperature range is from -4ºF (-20ºC) to 122ºF (50ºC). Air pressure range is from 700 hPa to 1060 hPa (10.2 psi to 15.4 psi). |
| Attitude Range |
|
| Insulin type | Rapid-acting U100 insulin (HumalogR and NovoRapidR) that has been prescribed by a healthcare professional. |
| Bolus delivery | Bolus Speeds
Programming Increments
Max bolus 25 units |
| Basal Rate Delivery | 0 to 35 units per hour. |
| Screen |
|
| Battery & Power | The pump requires one new AA (1.5V) battery. For best results, use a new AA lithium (FR6) battery. The pump also accepts an AA alkaline (LR6) or a fully charged AA NiMH (HR6) nickel-metal hydride rechargeable battery. |
| Pump memory | At any time you can review 35 days of pump history. |
| Size | The pump dimensions in centimeters are no greater than 10.2 length x 5.8 width x 2.8 depth. The mass of the insulin pump without battery and consumables is less than 117 grams. |
| Warranty |
|
| Compatible products | |
|---|---|
| Reservoir | Medtronic reservoir MMT-332A and MMT-342, 3.0 ml (300-unit), Medtronic reservoir MMT0326A, 1.8 ml (180-units) |
| Infusion Sets | Medtronic Diabetes offers a wide range of infusion sets so that you can choose the right one for your comfort and safety. https://www.medtronic-diabetes.com/en-ZA/insulin-pump-therapy/infusion-sets |
| Guardian™ Link 4 Transmitter | Used with your pump for Continuous Glucose Monitoring (CGM). A device that connects to a glucose sensor. The transmitter collects data measured by the sensor and wirelessly sends this data to monitoring devices. |
| Guardian™ 4 Sensor & transmitter | Used with your pump for Continuous Glucose Monitoring (CGM). Does not require finger pricks for calibration or to make diabetes treatment |
| Accessories | There are many ways to wear your insulin pump. You can attach your pump to your waistband or belt, protect it in your pocket, or even clip it under clothing. See our collection of accessories here. https://www.medtronic-diabetes.com/en-ZA/support/accessories |
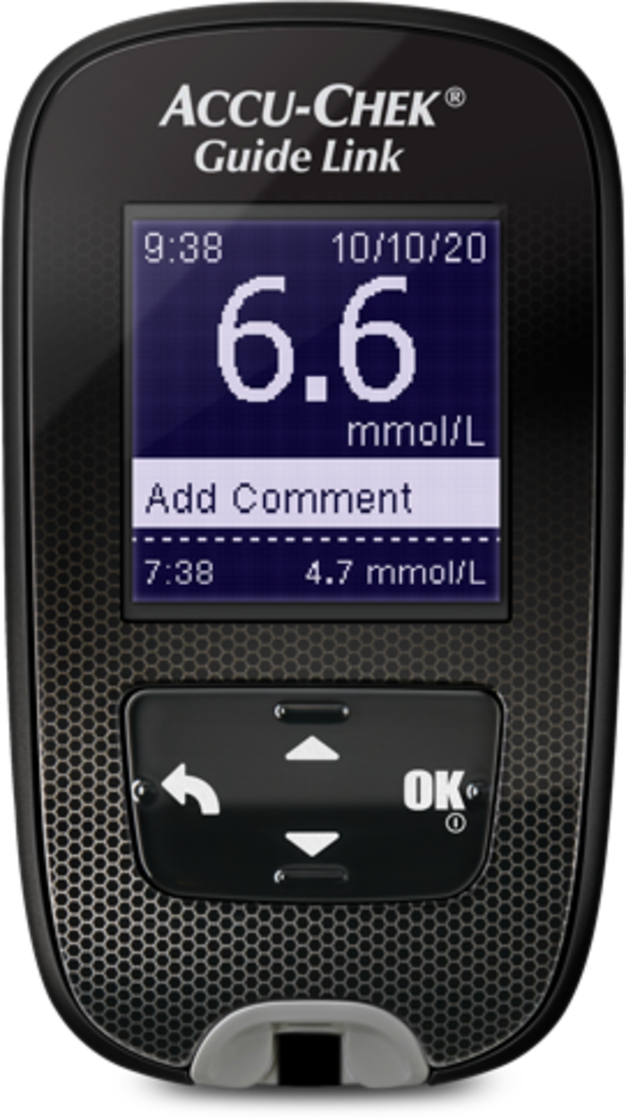
| Accu-Chek® Guide Blood Glucose Meter | The MiniMed™ 780G system comes with a compatible meter. It wirelessly connects to your pump, allowing you to send blood glucose meter readings to your |
App compatibility
| iOS | 13.0, 13.1, 13.2, 13.3, 13.5.1, 13.6.1, 13.7, 14.0, 14.0.1, 14.1, 14.2, 14.2.1, 14.3, 14.4, 14.5, 14.6, 14.7, 14.8, 15.0, 15.1, 15.2, 15.3, 15.4, 15.5, 15.6, 15.7, 16.0, 16.0.3, 16.1 |
| iPhone | SE (2020), iPod Touch (7th generation), 6S, 6S Plus, 7, 7 Plus, 8, 8 Plus, X, XS, XS Max, XR, 11 - (only OS 13 +), 11 Pro - (only OS 13 +), 11 Pro Max - (only OS 13 +), 12 - (only OS 14 +), 12 Mini - (only OS 14 +), 12 Pro - (only OS 14 +), 12 Pro Max - (only OS 14 +), 13, 13 Mini, 13 Pro, 13 Pro Max, 14, 14 Plus, 14 Pro, 14 Pro Max |
| Manufacturer | Model | Android OS |
|---|---|---|
| Samsung |
| Android OS: 9 |
| Samsung |
| Android OS: 9,10 |
| Samsung |
| Android OS: 9 |
| Samsung |
| Android OS: 10 |
| Samsung |
| Android OS: 10 |
| Samsung |
| Android OS: 9,10,11,12 |
| Samsung |
| Android OS: 10,11,12 |
| Samsung |
| Android OS: 11,12 |
| Samsung |
| Android OS: 11,12 |
| Samsung |
| Android OS: 11,12 |
| Samsung |
| Android OS: 12,13 |
| Samsung |
| Android OS: 9 |
| Android OS: 10 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12 | |
| Motorola |
| Android OS: 9 |
| Android OS: 9,10 | |
| Huawei |
| Android OS: 9,10 |
| One Plus |
| Android OS: 9,10 |
| Android OS: 12 | |
| Android OS: 12 | |
| LG |
| Android OS: 10 |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12,13 | |
| Android OS: 11,12,13 | |
| Sony™ |
| Android OS: 10 |
| Xiaomi |
| Android OS: 9,10 |
| Android OS: 12 |
| iOS | 13.0, 13.3.1, 13.4.1, 13.5.1, 13.6, 13.6.1, 13.7, 14.0, 14.0.1, 14.1, 14.2, 14.2.1, 14.4, 14.5, 14.6, 14.7, 14.8, 15.0, 15.1, 15.2, 15.3, 15.4, 15.5, 15.6, 15.7, 16.0 |
| iPhone | SE (2020), 6S, 6S Plus, 7, 7 Plus, 8, 8 Plus, X, XS, XS Max, XR, 11 - (only OS 13 +), 11 Pro - (only OS 13 +), 11 Pro Max - (only OS 13 +), 12 - (only OS 14 +), 12 Mini - (only OS 14 +), 12 Pro - (only OS 14 +), 12 Pro Max – (only OS 14 +), 13, 13 Mini, 13 Pro, 13 Pro Max, 14, 14 Plus, 14 Pro, 14 Pro Max |
| Manufacturer | Model | Android OS |
|---|---|---|
| Samsung |
| Android OS: 9 |
| Samsung |
| Android OS: 9,10 |
| Samsung |
| Android OS: 9,10,11 |
| Samsung |
| Android OS: 10,11,12 |
| Samsung |
| Android OS: 11,12,13 |
| Samsung |
| Android OS: 12,13 |
| Samsung |
| Android OS: 9 |
| Android OS: 10 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| Android OS: 10,11 | |
| LG™ |
| Android OS: 9 |
| Android OS: 10 | |
| Sony™ |
| Android OS: 10 |
| Android OS: 10,11,12 | |
| Android OS: 10,11,12,13 | |
| Android OS: 11,12,13 | |
| Huawei |
| Android OS: 9,10 |
| Xiaomi |
| Android OS: 9,10 |
| Android OS: 12 | |
| OnePlus |
| Android OS: 10, 11 |
| Android OS: 12 | |
| Android OS: 12 |
Frequently asked questions
The MiniMedTM 780G system is approved for people with type 1 diabetes, age 7 years and above. If you would like a CGM and not an insulin pump, consider looking into Smart MDI System.
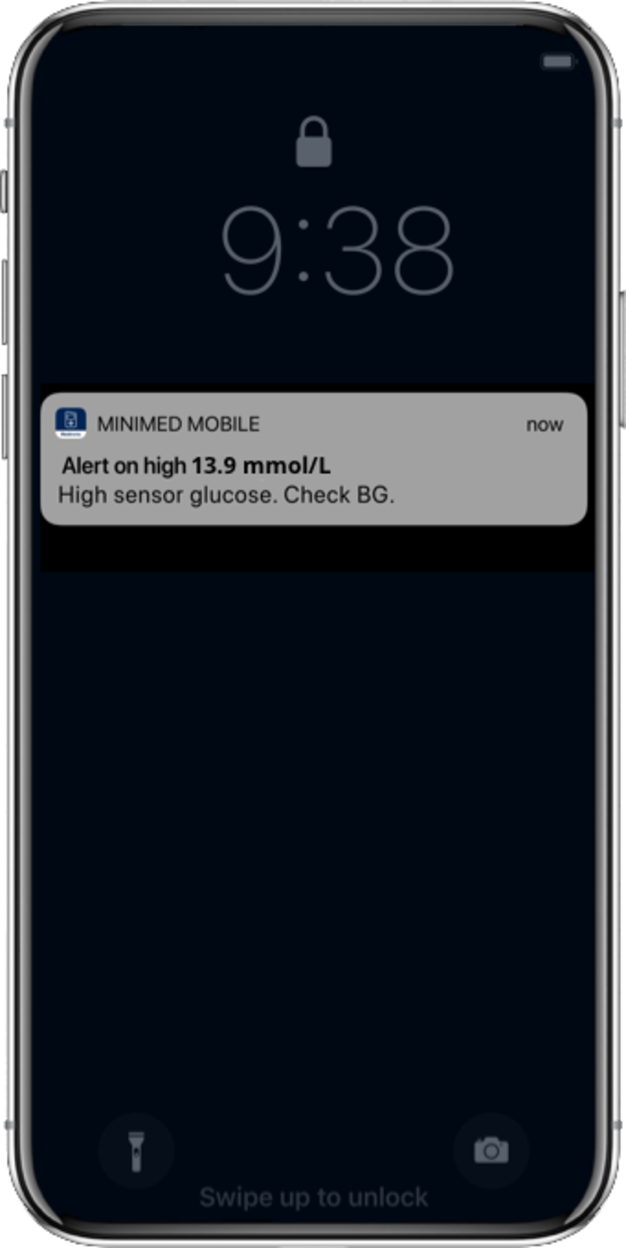
Yes. With the MiniMed™ Mobile app that connects with the MiniMed™ 780G system, you can easily track your glucose levels and view notifications and alerts on your smartphone. Up to 5 care-partners can be invited to download the CareLink™ Connect app and view your real-time glucose values on their phones. Available to download at no cost for most iOS and Android smartphones. To see if your smartphone is compatible, see this reference chart.
The MiniMed™ 780G system automatically adjust insulin delivery to your needs for an easier way to stabilise glucose
The MiniMed™ 780G system, automatically adjust delivery of basal insulin based on Continuous Glucose Monitoring (CGM) values. This means that the pump gets glucose readings from the Guardian™ Sensor 4 and transmitter automatically, and then delivers of a variable rate of insulin, 24 hours a day based on your personal needs. This integrated system – meaning having a pump and a CGM that speak to one another – can help to stabilise your glucose levels and reduce both high and low glucose levels.
When you use an insulin pump, you have a dramatic reduction in injections – by up to 90%.* The infusion set that attaches to your body contains a small, flexible plastic cannula that is inserted to be placed under the skin to allow for continuous infusion of insulin. It is changed every 2 – 3 days. A CGM glucose sensor is inserted every 7 days.
Using the SmartGuard™ feature does not completely replace finger pricks but you will likely check your BG less frequently. Sensor readings are taken from your interstitial fluid (fluid surrounding the cells in your tissue) and not your blood, so you still need to test using a BG metre for BG readings. The system may require a BG reading to continue using the SmartGuard™ feature. It is important to know that all blood glucose values entered & accepted are used for calibration.
* Assumes four injections per day for 30 days and one infusion set change every two to three days.
- Get started by clicking on “Request a call back” or contacting us https://www.medtronic-diabetes.com/en-ZA/contact-medtronic-diabetes so we can have someone follow up with you. We will help answer any questions, discuss out-of-pocket costs, if you plan to self-fund.
- Once the system is purchased and shipped, we will work with you to schedule your training. We have a strong training team who will conduct all the training you need. With the use of CGM and SmartGuardTM technology, you may need a few training sessions.
- Once you have been trained, we will continue to support you throughout your journey with us. We will provide ongoing educational resources and also offer a dedicated Technical Support team who are available 24/7 0800 633 7867.
If you are a current customer, contact your local representative or find our contact details here https://www.medtronic-diabetes.com/en-ZA/contact-medtronic-diabetes to discuss your options for upgrading.
If you are a healthcare professional we encourage you to visit https://hcp.medtronic-diabetes.com/en-GB
- Choudhary P, et al. Diabetes Technol Ther 2024; 26 (Suppl. 3): S32-S37.
- De Meulemeester J. The association of chronic complications with time in tight range and time in range in people with type 1 diabetes: a retrospective cross-sectional real-world study. Diabetologia. 2024;67(8):1527-1535. doi:10.1007/s00125-024-06171-y
- Petrovski G, et al. Diabetes Care 2023;46(3):544–550
- Di Molfetta S, Di Gioia L, Caruso I, et al. Efficacy and Safety of Different Hybrid Closed Loop Systems for Automated Insulin Delivery in People With Type 1 Diabetes: A Systematic Review and Network Meta-Analysis. Diabetes Metab Res Rev. 2024;40(6):e3842. doi:10.1002/dmrr.3842
- dQ&A Diabetes Technology Report, 2024 (n= 2,721)
- Fusselman H, et al. FUSS2015D-P - The Extended Wear Infusion Set - A Design for Plastic Waste Reduction. Virtual Diabetes Technology Meeting. 2020
- Arrieta A, et al. Diabetes Obes Metab. 2022;24(7):1370-1379. Mean 80% TIR is reached using the recommended optimal settings of 2 hours active insulin time and 100 mg/dL (5.5 mmol/L) as glucose target for at least 90% of the time.
- * Compared to MDI + isCGM
- ** Refer to System User Guide - SmartGuard™ feature. Some user interaction required
- ^ A blood glucose (BG) reading is needed when entering SmartGuard™ feature. If glucose alerts and CGM readings do not match your symptoms, use a BG meter to make diabetes treatment decisions. Refer to System User Guide - SmartGuard™ feature. Some user interaction required.
- ^^ Multiple Daily Injections (3 bolus and 1 basal insulin per day) requires 28 injections per week vs. 1 with Medtronic Extended Infusion Set.
- # Based on RWE publications.
- ~ The people with diabetes testimonials relates to the account of an individual's response to the treatment. The account is genuine, typical and documented. However, the individual's response does not provide any indication, guide, warranty or guarantee as to the response other people with diabetes may have to the treatment. The response other people with diabetes have to the treatment could be different. Responses to the treatment discussed can and do vary and specific to the individual people with diabetes.
- ¥ Refer to Accu-Chek® Guide Link User's Manual
- 1.Choudhary P, et al. Diabetes Technol Ther 2024; 26 (Suppl. 3): S32-S37.
- 2.De Meulemeester J. The association of chronic complications with time in tight range and time in range in people with type 1 diabetes: a retrospective cross-sectional real-world study. Diabetologia. 2024;67(8):1527-1535. doi:10.1007/s00125-024-06171-y
- 3.Petrovski G, et al. Diabetes Care 2023;46(3):544–550
- 4.Di Molfetta S, Di Gioia L, Caruso I, et al. Efficacy and Safety of Different Hybrid Closed Loop Systems for Automated Insulin Delivery in People With Type 1 Diabetes: A Systematic Review and Network Meta-Analysis. Diabetes Metab Res Rev. 2024;40(6):e3842. doi:10.1002/dmrr.3842
- 5.dQ&A Diabetes Technology Report, 2024 (n= 2,721)
- 6.Fusselman H, et al. FUSS2015D-P - The Extended Wear Infusion Set - A Design for Plastic Waste Reduction. Virtual Diabetes Technology Meeting. 2020
- 7.Arrieta A, et al. Diabetes Obes Metab. 2022;24(7):1370-1379. Mean 80% TIR is reached using the recommended optimal settings of 2 hours active insulin time and 100 mg/dL (5.5 mmol/L) as glucose target for at least 90% of the time.
- 1.Choudhary P, et al. Diabetes Technol Ther 2024; 26 (Suppl. 3): S32-S37.
- 2.De Meulemeester J. The association of chronic complications with time in tight range and time in range in people with type 1 diabetes: a retrospective cross-sectional real-world study. Diabetologia. 2024;67(8):1527-1535. doi:10.1007/s00125-024-06171-y
- 3.Petrovski G, et al. Diabetes Care 2023;46(3):544–550
- * Compared to MDI + isCGM
- ** Refer to System User Guide - SmartGuard™ feature. Some user interaction required
- ^ A blood glucose (BG) reading is needed when entering SmartGuard™ feature. If glucose alerts and CGM readings do not match your symptoms, use a BG meter to make diabetes treatment decisions. Refer to System User Guide - SmartGuard™ feature. Some user interaction required.
- ^^ Multiple Daily Injections (3 bolus and 1 basal insulin per day) requires 28 injections per week vs. 1 with Medtronic Extended Infusion Set.
- # Based on RWE publications.
- ~ The people with diabetes testimonials relates to the account of an individual's response to the treatment. The account is genuine, typical and documented. However, the individual's response does not provide any indication, guide, warranty or guarantee as to the response other people with diabetes may have to the treatment. The response other people with diabetes have to the treatment could be different. Responses to the treatment discussed can and do vary and specific to the individual people with diabetes.
- ¥ Refer to Accu-Chek® Guide Link User's Manual
- ** Refer to System User Guide - SmartGuard™ feature. Some user interaction required
- ^ A blood glucose (BG) reading is needed when entering SmartGuard™ feature. If glucose alerts and CGM readings do not match your symptoms, use a BG meter to make diabetes treatment decisions. Refer to System User Guide - SmartGuard™ feature. Some user interaction required.
All clinical data and surveys results included in this document refer to MiniMed™ 780G system used in combination with Guardian™ sensor 3. Information contained herein is not medical advice and should not be used as an alternative to speaking with your doctor. Discuss indications, contraindications, warnings, precautions, potential adverse events and any further information with your health care professional.
UK-IPT-2300174©2023 Medtronic. All rights reserved Medtronic. Medtronic logo and Further, Together are trademarks of Medtronic. Third party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company. ACCU-CHEK AND ACCU-CHEK GUIDE LINK are trademarks of Roche Diabetes Care. DreaMed Diabetes is a trademark of DreaMed Diabetes. Ltd. The MiniMed™ 780G system algorithm includes technology developed by DreaMed Diabetes. The legal manufacturer of MiniMed™ Mio ™ Advance infusion set is Unomedical a/s. Aaholmvej 1-3, Osted. 4320 Lejre, Denmark.