MiniMed™ 780G systemLive life with more
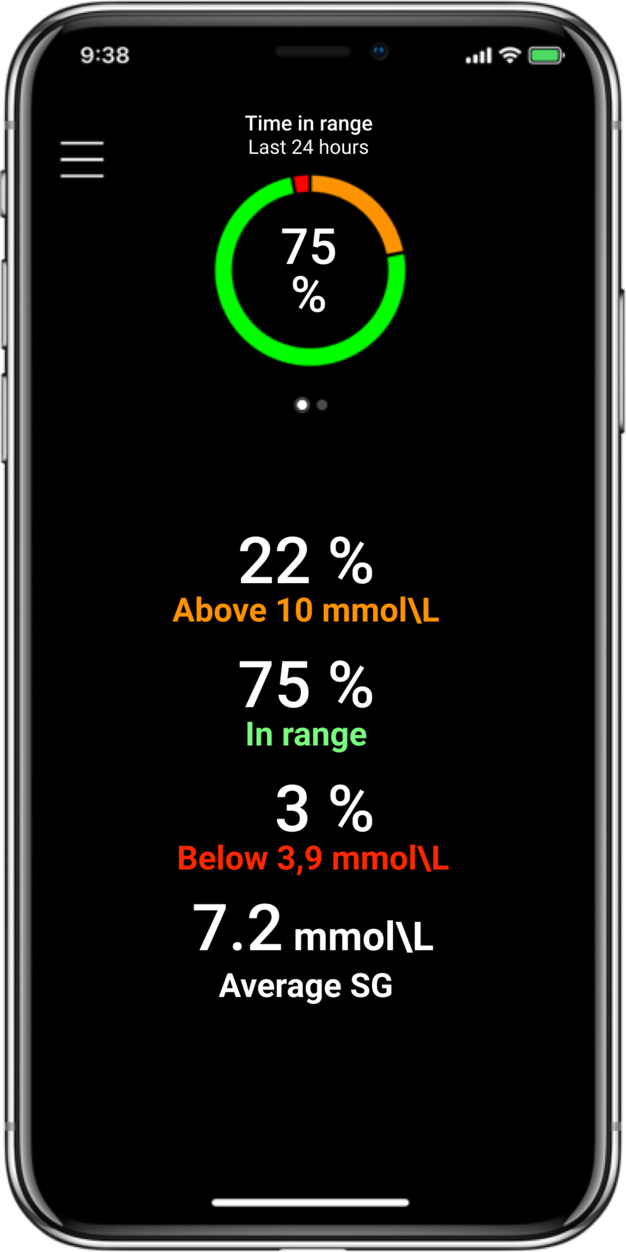
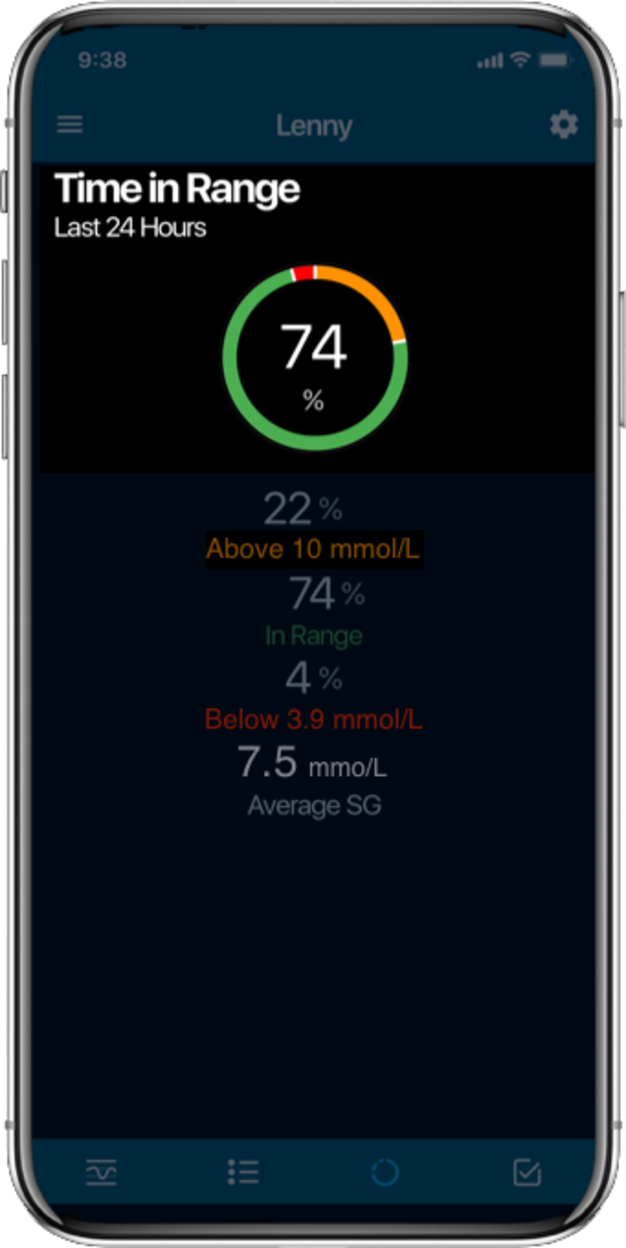
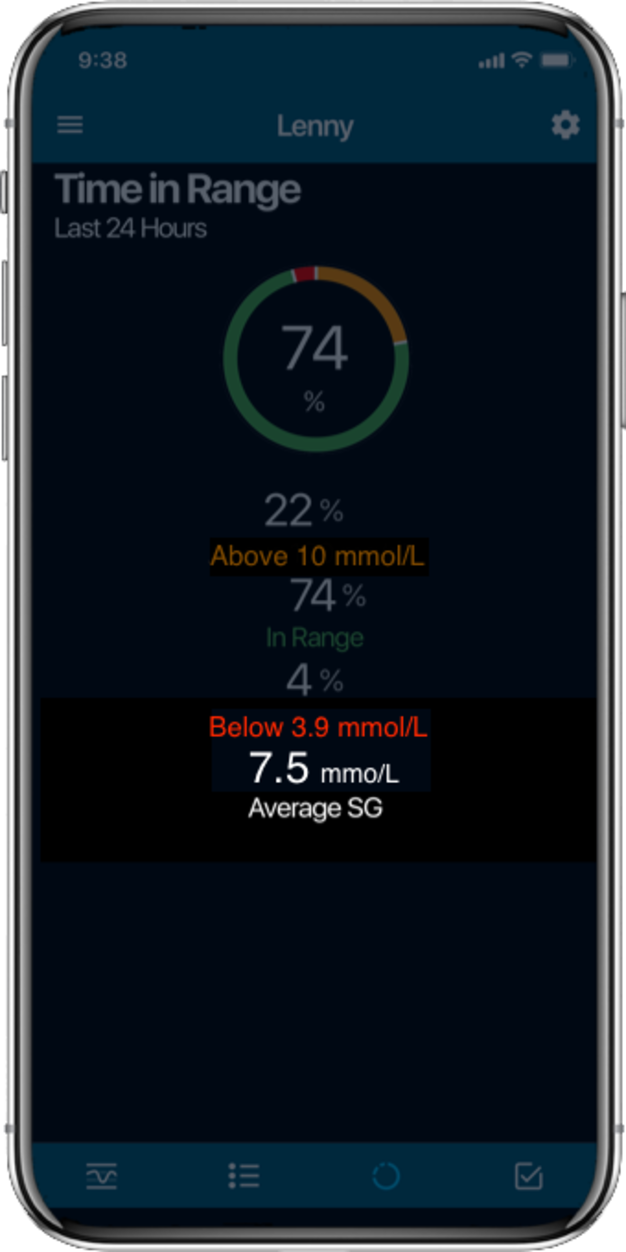
Time in Range1,
For people with type 1 diabetes
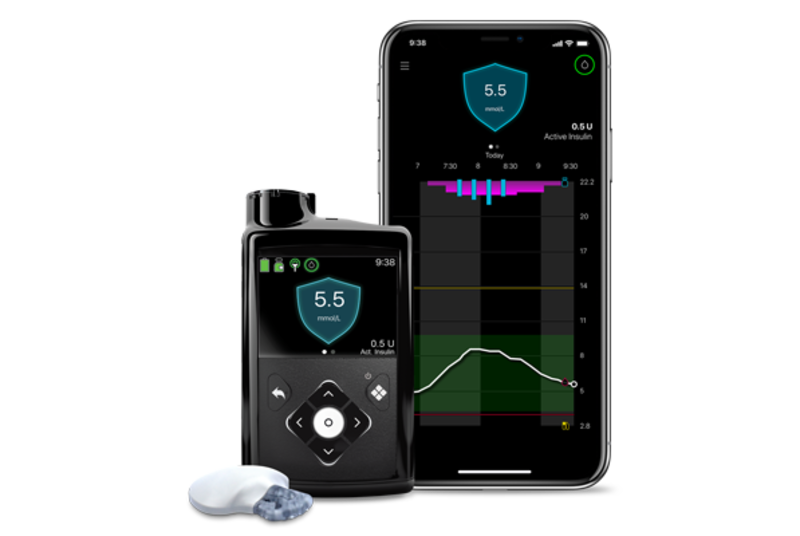
MiniMed™ 780G system
More Time in Range1, , day and night.
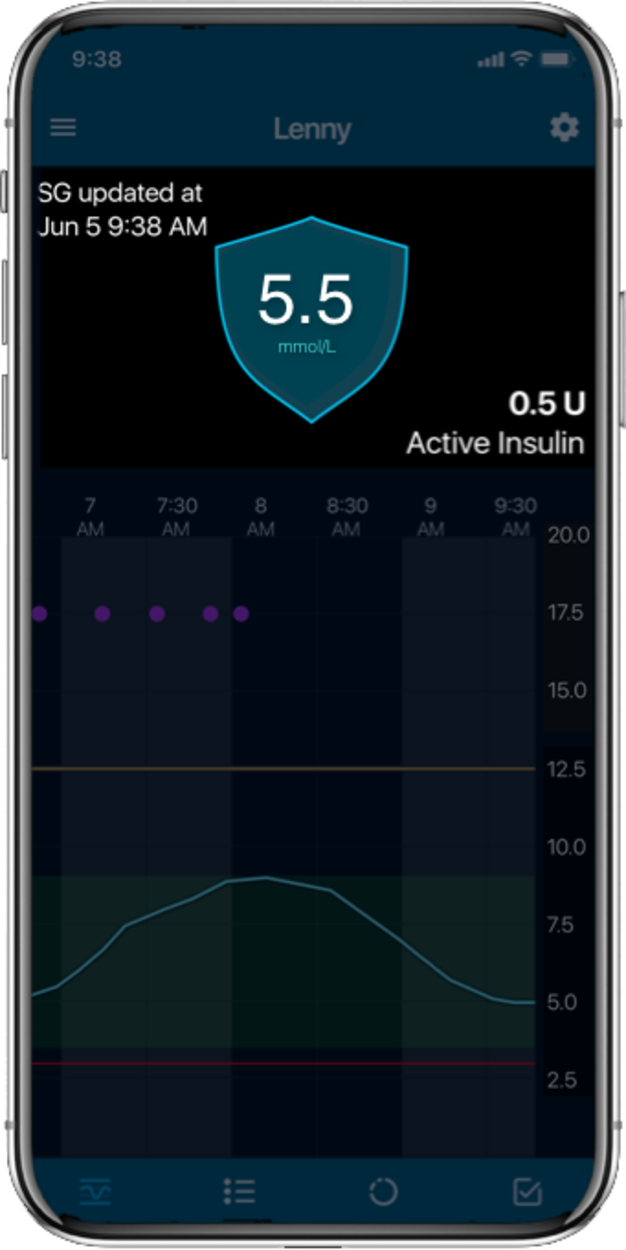
The MiniMed™ 780G system

Considering an automated insulin pump system?
When choosing an insulin pump, make sure it meets your needs and delivers the best results. Key considerations should include:
- Time in Range (TIR):
The percentage of time your blood glucose levels are within the target range. Higher TIR reduces complications like eye damage (retinopathy), heart and kidneydisease2 . - Burden reduction:
Achieve more while doing less. Your pump should help you to stay in range even when carb counting isn'texact3.
Things you should know about the MiniMed™ 780G
Advanced algorithm
The algorithm adapts to you providing better time in range than other
Wear what you want
Feel free to hide or show the pump depending on your outfit choice.
No daily charging
Unlike other pumps, the 780G doesn't need daily charging.
Freedom to disconnect
Disconnect for up to an hour for activities.
Waterproof design
A tough, easy-to-use pump.
Trusted across Europe
used by hundreds of thousands, MiniMed™ 780G is Europe's most popular automated insulin pump.
SmartGuard™ technology
Our most advanced insulin pump system
with SmartGuard™ technology
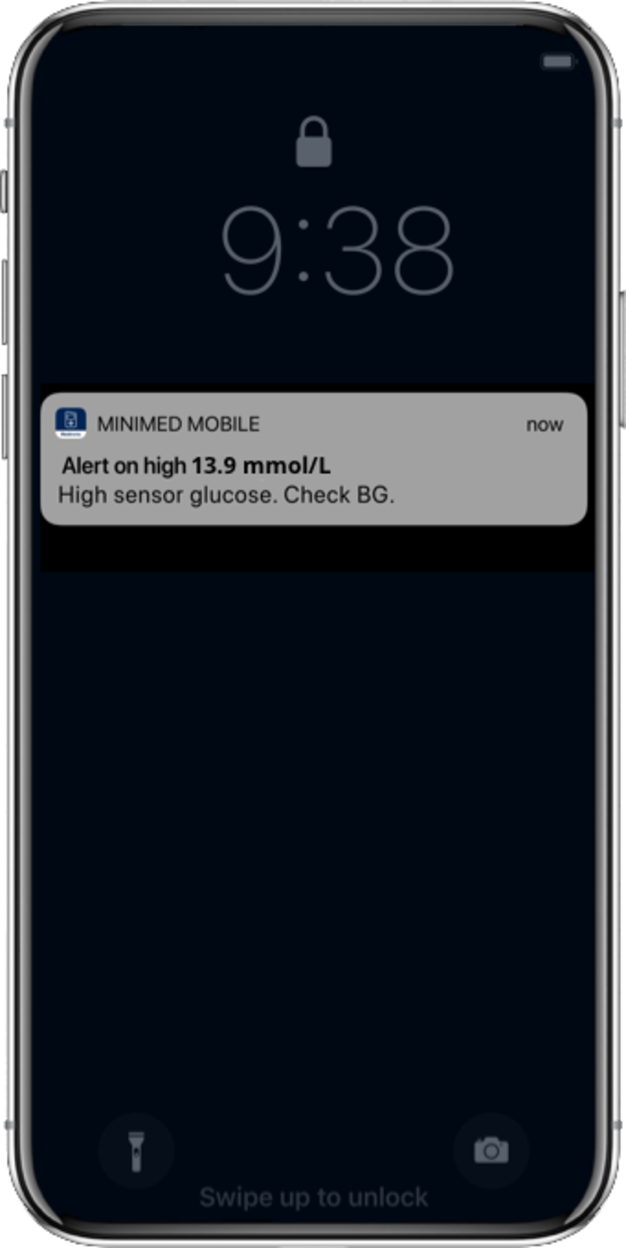
SmartGuard™ technology continuously anticipates your insulin needs, adjusts insulin delivery, and corrects highs
Meal detection
Stronger correction
bolus allowed while
meal is detected
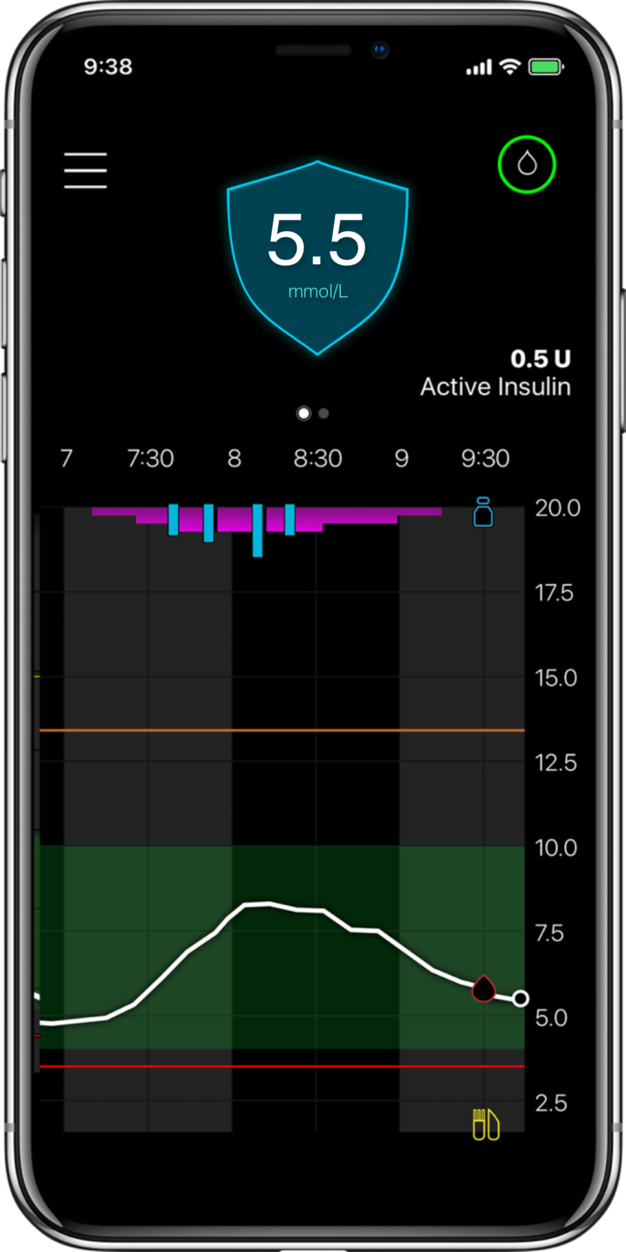
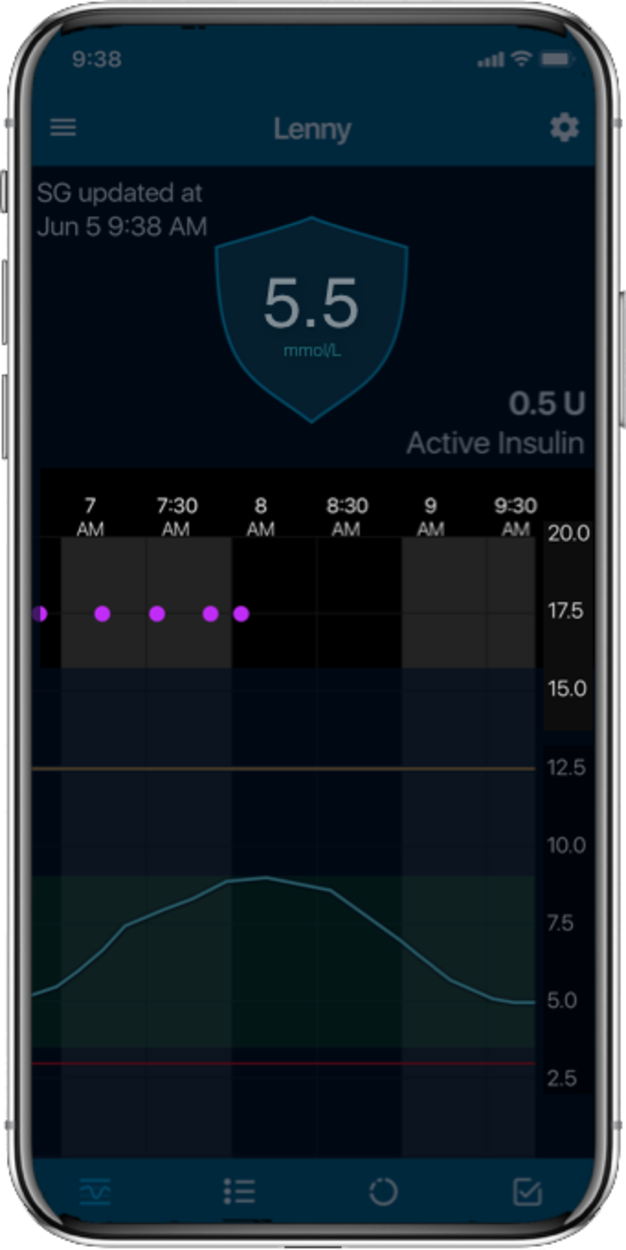
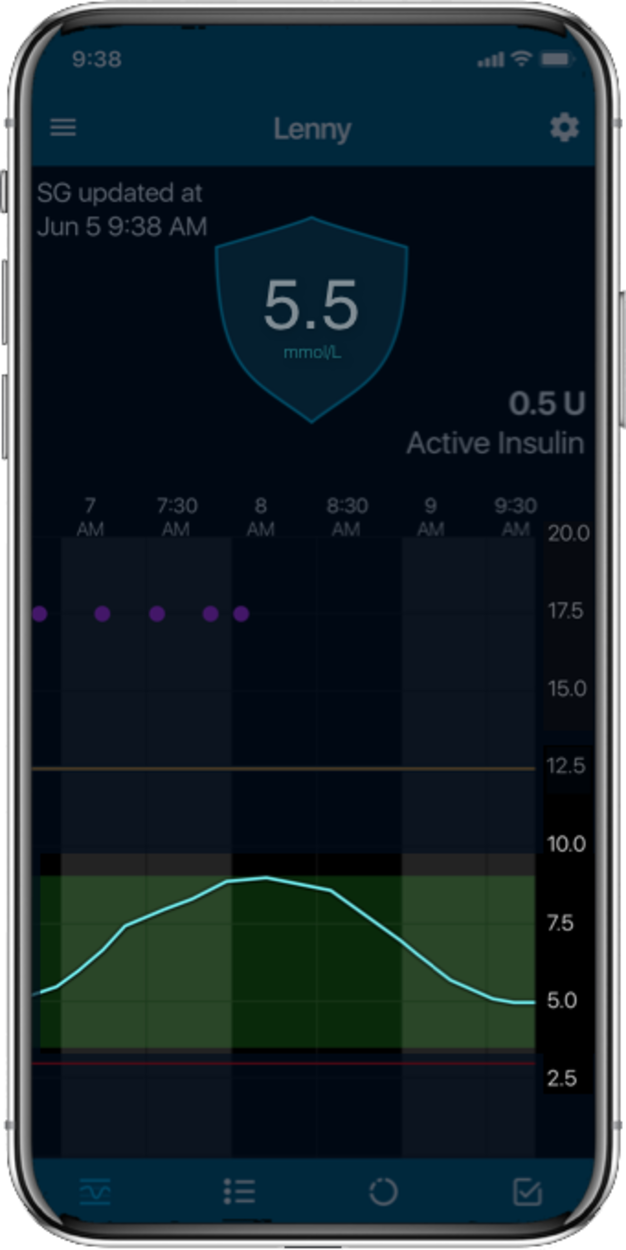
Glucose levels
Automatic adjustments
Auto corrections
Meal detection
For illustrative purposes only.
Compare MiniMed™ 780G system
It’s easy to see the difference between our system and our competitors.
Minimed™ 780G
Omnipod 5
Ypsomed
Tandem T-Slim
Higher TIR reduces complications like eye damage (retinopathy), heart and kidney
- μ Mean 81.3% TIR is reached using the recommended optimal settings of 2 hours active insulin time and 100 mg/dL (5.5 mmol/L) as glucose target for at least 90% of the time by users aged 15 and older.